Strontium is the chemical element with atomic number 38, symbol Sr
Strontium, like calcium, is an alkaline earth. It is soft, malleable, gray-yellow. On contact with air, it forms a protective oxide film (passivation). It ignites and burns easily in air and reacts with water.
It is found mainly in the minerals celestite and strontianite. China is now the leading producer of strontium. Strontium metal can be prepared by electrolysis of the molten strontium chloride and potassium chloride, or by reducing strontium oxide with aluminium in a vacuum.
Usage
Used for producing glass (cathode ray tubes) for color televisions. It is also used in producing ferrite ceramic magnets and in refining zinc.
Its chloride is used in the management of bone pain in prostate cancer.
Its salts are used in flares and fireworks for a crimson color.
Its chloride is used in toothpaste for sensitive teeth.
Its oxide is used to improve the quality of pottery glazes.
Modern ‘glow-in-the-dark’ paints and plastics contain strontium aluminate.
The isotope 90Sr is one of the best long-lived, high-energy beta emitters known. It is used in cancer therapy.
Health consequences
Water insoluble strontium compounds can become insoluble in water as a result of chemical reactions. Soluble compounds are more likely to affect our health than insoluble compounds because they can contaminate drinking water. Fortunately, the concentrations in drinking water are, in general, quite low.
You can be exposed to small amounts of (radioactive) strontium by breathing air or dust, eating, drinking, or in contact with soil containing of it. Usually exposure is through food or water.
Its concentrations in food contribute to the strontium concentration in the human body. Foods that contain high levels of strontium are seeds, leafy vegetables and dairy products.
Most people have it in moderate intake. The only its compound that is considered hazardous to health, even in small amounts, is strontium chromate. This dangerousness is mainly due to the toxic chromium it contains. Its chromate is known to cause lung cancer, but the risks of exposure have been greatly reduced by company safety procedures, so it is no longer a significant health risk.
Consumption of high concentrations of strontium is generally not known to be harmful to health. Only one case of strontium allergy was discovered, but there were no other similar cases. In children, consuming too much strontium can pose a risk as it can cause problems with bone growth.
When the consumption of strontium is extremely high, it can cause an interruption in bone development, but this only happens when the consumption is in the order of a thousand ppm. The level of strontium in food and drinking water is not high enough to cause this type of effect.
Radioactive strontium represents a greater health risk than stable strontium. When consumption is very high, it can cause anemia and oxygen deficiency and, at extremely high concentrations, it is even known to cause cancer due to the alteration of the genetic material of cells.
Environmental impact
Strontium in its elemental form is naturally present in the environment (rocks, soil, water, air). Its compounds can move around the environment quite easily because many of them are soluble.
It is always present in the air as dust up to a certain level. Its concentrations in the air are increased by human activities, such as the combustion of coal and oil. Dust particles that contain strontium can be deposited in surface water, soil and on plants. When particles do not settle, they fall back to the earth’s surface during rain or snowfall. All the strontium ends up in the soil or the bottom of surface water, where it mixes with the strontium already present.
It can also infiltrate water after being in soils. Only a small part of the strontium in water comes from dust particles in the air. Most of the strontium in the water is dissolved, but some remains in suspension, resulting in the formation of muddy water. Little strontium is found in drinking water.
When its concentrations in water exceed regular concentrations, it is usually caused by human activities, mainly when wastes are released directly into water. These excessive concentrations can also be caused by the deposition of air dust which reacts with strontium particles emitted during industrial processes.
The increase in soil concentrations can also be favored by human activities such as the discharge of coal or incinerator ash. Strontium in soil dissolves in water, therefore, it is likely to travel deeper in the soil and join groundwater. Some of the strontium introduced by humans does not reach surface water and can remain in the soil for decades.
Due to the nature of it, some of them can be taken up by fish, vegetables, livestock and other animals.
One of the isotopes of strontium is radioactive. This isotope is a priori not naturally present in the environment. It is there because of man: nuclear bomb tests, leaks of radioactive storage. The only way to decrease the concentrations of this isotope is the radioactive decay to stable zirconium.
Concentrations of radioactive strontium in the environment are relatively low, and the particles always end up in the soil or on the ocean floor, where they mix with other strontium particles. Radioactive strontium is not likely to end up in drinking water.
Strontium in the periodic table
| Atomic number (Z) | 38 |
|---|---|
| Group | group 2 (alkaline earth metals) |
| Period | period 5 |
| Block | s-block |
| Electron configuration | [Kr] 5s2 |
| Electrons per shell | 2, 8, 18 |
Physical properties
| Physical properties | |
|---|---|
| Phase at STP | solid |
| Melting point | 1050 K (777 °C, 1431 °F) |
| Boiling point | 1650 K (1377 °C, 2511 °F) |
| Density (near r.t.) | 2.64 g/cm3 |
| when liquid (at m.p.) | 2.375 g/cm3 |
| Heat of fusion | 7.43 kJ/mol |
| Heat of vaporization | 141 kJ/mol |
| Molar heat capacity | 26.4 J/(mol·K) |
Vapor pressure
| P (Pa) | 1 | 10 | 100 | 1 k | 10 k | 100 k |
|---|---|---|---|---|---|---|
| at T (K) | 796 | 882 | 990 | 1139 | 1345 | 1646 |
Atomic properties
| Atomic properties | |
|---|---|
| Oxidation states | +1, +2 (a strongly basic oxide) |
| Electronegativity | Pauling scale: 0.95 |
| Ionization energies |
|
| Atomic radius | empirical: 215 pm |
| Covalent radius | 195±10 pm |
| Van der Waals radius | 249 pm |
Other properties
| Other properties | |
|---|---|
| Natural occurrence | primordial |
| Crystal structure | face-centered cubic (fcc) |
| Thermal expansion | 22.5 µm/(m·K) (at 25 °C) |
| Thermal conductivity | 35.4 W/(m·K) |
| Electrical resistivity | 132 nΩ·m (at 20 °C) |
| Magnetic ordering | paramagnetic |
| Magnetic susceptibility | −92.0·10−6 cm3/mol (298 K) |
| Young’s modulus | 15.7 GPa |
| Shear modulus | 6.03 GPa |
| Poisson ratio | 0.28 |
| Mohs hardness | 1.5 |
| CAS Number | 7440-24-6 |
History
In 1787, an unusual rock which had been found in a lead mine at Strontian, Scotland, was investigated by Adair Crawford, an Edinburgh doctor. He realised it was a new mineral containing an unknown ‘earth’ which he named strontia. In 1791, another Edinburgh man, Thomas Charles Hope, made a fuller investigation of it and proved it was a new element. He also noted that it caused the flame of a candle to burn red.
Meanwhile Martin Heinrich Klaproth in Germany was working with the same mineral and he produced both strontium oxide and strontium hydroxide.
Strontium metal itself was isolated in 1808 at the Royal Institution in London by Humphry Davy by means of electrolysis, using the method with which he had already isolated sodium and potassium.
| History | |
|---|---|
| Naming | after the mineral strontianite, itself named after Strontian, Scotland |
| Discovery | William Cruickshank (1787) |
| First isolation | Humphry Davy (1808) |
Main isotopes
| Main isotopes of strontium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Periodic Table of Elements | Complete List of Chemical Elements by Group, Name, Symbol, Color and Type
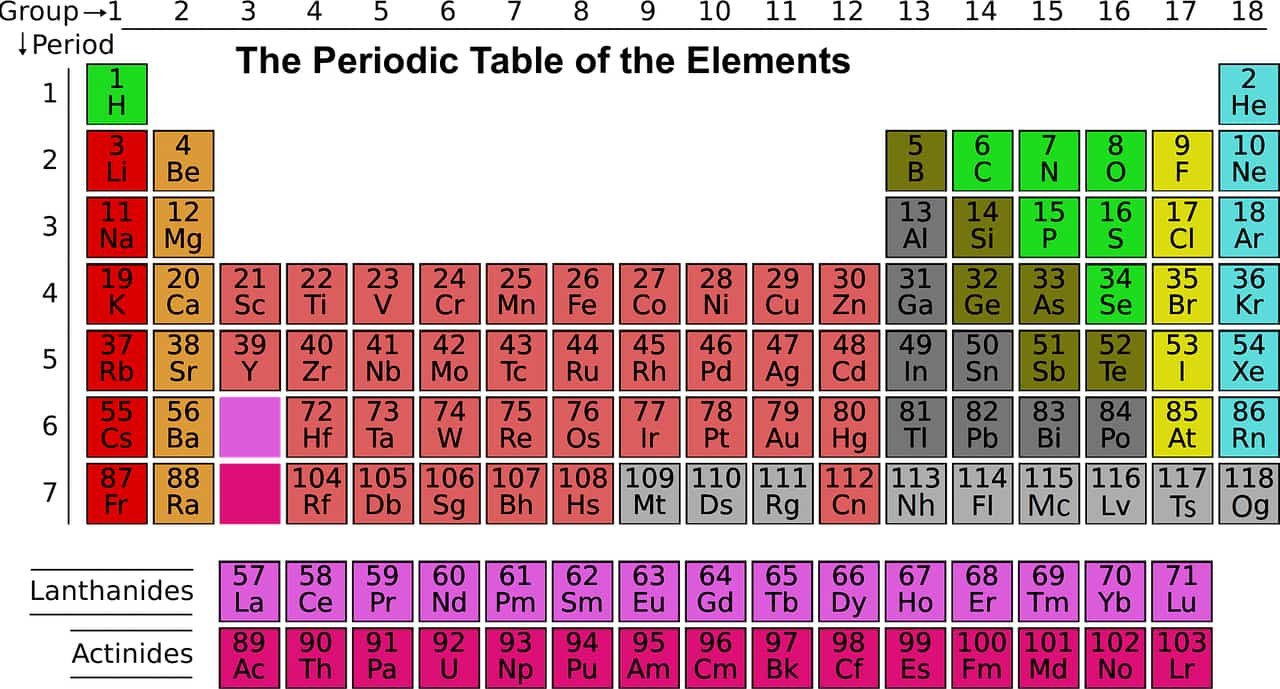
Sources: PinterPandai, Live Science, Royal Society of Chemistry
Photo credit: Wikimedia Commons
Photo explanations: The chemical element strontium as synthetic crystals, sealed under argon in a glass ampoule, purity (99.95 %). Size of the image ca. 3cm * 4,7cm.