Barium is the chemical element with atomic number 56, symbol Ba
Barium oxide was discovered in 1774 by Carl Wilhelm Scheele in manganese dioxide ores. It was isolated in 1808 by the Englishman Sir Humphry Davy, then purified by Antoine Guntz in 1901. The word barium was coined from the Greek βαρύς (“heavy”).
Uses
Barium is used pure for trapping residual gases in cathode-ray tubes or as an indicator of the presence of air in evacuated tube solar thermal collectors.
Small amounts of salts of this element (acetate, carbonate, chlorate, chloride, hydroxide, nitrate, oxide, perchlorate, peroxide, polysulfide, sulfate or sulfide) are used in many fabrications, including:
- photographic paper,
- high temperature resistant lubricant;
- in glasses, ceramics, enamels and porcelains and their varnishes and glazes, barium silicates are produced by adding barium compounds, examples:
- BaSO4 + SiO2 → BaSiO3 + SO3 (très haute température)
- BaCO3 + SiO2 → BaSiO3 + CO2
- Ba(NO3)2 + SiO2 → BaSiO3 + N2O5
- BaO + SiO2 → BaSiO3,
In glass, it increases the absorption of X and gamma rays as well as the refractive index without increasing the chromatic aberration too much,
at low temperature in a glaze, barium sulphate can precipitate in the form of large sparkling crystals or small opaque crystals. It is also used to adjust the coefficient of expansion or the viscosity by replacing other oxides;
in pyrotechnics, barium colors the flame apple green, nitrate and perchlorate are preferred because they provide the oxidant.
As an opacifier in radiology. In this indication, given the strong toxicity in the dissolved state, sulphate is used, which is insoluble even in the acidic environment of the stomach;
sulphate is also used in drilling muds for its density;
Titanate (BaTiO3) serves as the basis for piezoelectric ceramics. Barium titanate is used, among other things, for the insulating part of certain capacitors.
It is a constituent element of nonlinear crystals (barium beta borate), used in particular to produce frequency mixtures in optics (nonlinear optics);
Painting
- in the form of barium sulphate or fixed white, it is used as a filler for paints and varnishes.
lithopone is the insoluble pigment of barium sulphate and zinc sulphide produced by mixing aqueous solutions of barium sulphide and zinc sulphate. - barium and copper silicates were made in China from the Zhou West period (1045-771 BC) to the Han period (AD 220) as pigments: Han blue (BaCuSi4O10) and Han purple (BaCuSi2O6).
Health effects of Barium
The levels of barium naturally present in the environment are very low. Large amounts of barium can only be found in soils and foods such as nuts, seaweed, fish and some plants. The amount of barium that is detected in food or water is usually not high enough to become a health concern.
People with a greater risk of exposure to barium, with health consequences, are those who work in the barium industry. Most of the health problems they can experience are caused by breathing air that contains barium sulfate or calcium carbonate.
Many hazardous waste sites contain some amount of barium. People living nearby can be exposed to harmful levels. This can lead to exposure by breathing dust, eating plants or soil, or drinking water polluted with it. There may also be contact with the skin.
The health consequences depend on the water solubility of the compounds. It compounds that dissolve in water can be harmful to health. The absorption of a large amount of these compounds can cause paralysis and, in some cases, death.
Small amounts of these compounds can cause breathing difficulties, increased blood pressure, changes in heart rate, stomach irritation, muscle weakness, changes in nerve reflexes, inflammation of the brain and liver, kidney and heart damage.
It has not been shown to cause cancer in humans. There is no evidence that it can cause infertility or birth problems.
Effects of barium on the environment
It is a silvery white metal that can be found in the environment, where it occurs naturally. It is present there in combination with other chemicals such as sulfur, carbon and oxygen.
It compounds are used by the oil and gas industries in drilling muds. Drilling muds simplify drilling in rocks by lubricating the drill rig.
It compounds are also used to make paints, bricks, tiles, glass and rubber.
Due to the intensive use of barium in industry, significant amounts of barium have been added to the environment. Therefore, in some places, the concentrations of barium in air, water and soil may be higher than the concentrations that are found naturally.
It enters the air during mining and refining processes, and during the production of it compounds. It can also be found in the air during the combustion of coal and oil.
Some of their compounds that are released in industrial processes easily dissolve in water and can be found in lakes, rivers and streams. Since they are soluble in water, these compounds can travel long distances. When fish and other aquatic organisms absorb barium compounds, barium builds up in their bodies. It compounds that are persistent generally remain on the surface of soils or in sediments at the bottom of the water. In general, low levels of barium are found in soils, these levels may be higher at hazardous waste sites.
Barium in the periodic table
| Atomic number (Z) | 56 |
|---|---|
| Group | group 2 (alkaline earth metals) |
| Period | period 6 |
| Block | s-block |
| Electron configuration | [Xe] 6s2 |
| Electrons per shell |
Physical Properties
Soft and silvery, barium is found in nature in the form of minerals, especially in the company of magnesium, manganese, calcium or strontium. As it reacts strongly with oxygen, it cannot be found as a native element.
| Physical properties | |
|---|---|
| Phase at STP | solid |
| Melting point | 1000 K (727 °C, 1341 °F) |
| Boiling point | 2118 K (1845 °C, 3353 °F) |
| Density (near r.t.) | 3.51 g/cm3 |
| when liquid (at m.p.) | 3.338 g/cm3 |
| Heat of fusion | 7.12 kJ/mol |
| Heat of vaporization | 142 kJ/mol |
| Molar heat capacity | 28.07 J/(mol·K) |
History of barium
It was discovered in oxide form by Scheele in 1774, then Davy isolated impure barium in 1808. The isolation of pure barium is due to Guntz in 1901.
| History | |
|---|---|
| Discovery | Carl Wilhelm Scheele (1772) |
| First isolation | Humphry Davy (1808) |
Vapor pressure
| P (Pa) | 1 | 10 | 100 | 1 k | 10 k | 100 k |
|---|---|---|---|---|---|---|
| at T (K) | 911 | 1038 | 1185 | 1388 | 1686 | 2170 |
Atomic properties
| Atomic properties | |
|---|---|
| Oxidation states | +1, +2 (a strongly basic oxide) |
| Electronegativity | Pauling scale: 0.89 |
| Ionization energies |
|
| Atomic radius | empirical: 222 pm |
| Covalent radius | 215±11 pm |
| Van der Waals radius | 268 pm |
Other properties
| Other properties | |
|---|---|
| Natural occurrence | primordial |
| Crystal structure | body-centered cubic (bcc) |
| Speed of sound thin rod | 1620 m/s (at 20 °C) |
| Thermal expansion | 20.6 µm/(m·K) (at 25 °C) |
| Thermal conductivity | 18.4 W/(m·K) |
| Electrical resistivity | 332 nΩ·m (at 20 °C) |
| Magnetic ordering | paramagnetic |
| Magnetic susceptibility | +20.6·10−6 cm3/mol |
| Young’s modulus | 13 GPa |
| Shear modulus | 4.9 GPa |
| Bulk modulus | 9.6 GPa |
| Mohs hardness | 1.25 |
| CAS Number | 7440-39-3 |
Main isotopes
| Main isotopes of barium | ||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||
/
Periodic Table of Elements | Complete List of Chemical Elements by Group, Name, Symbol, Color and Type
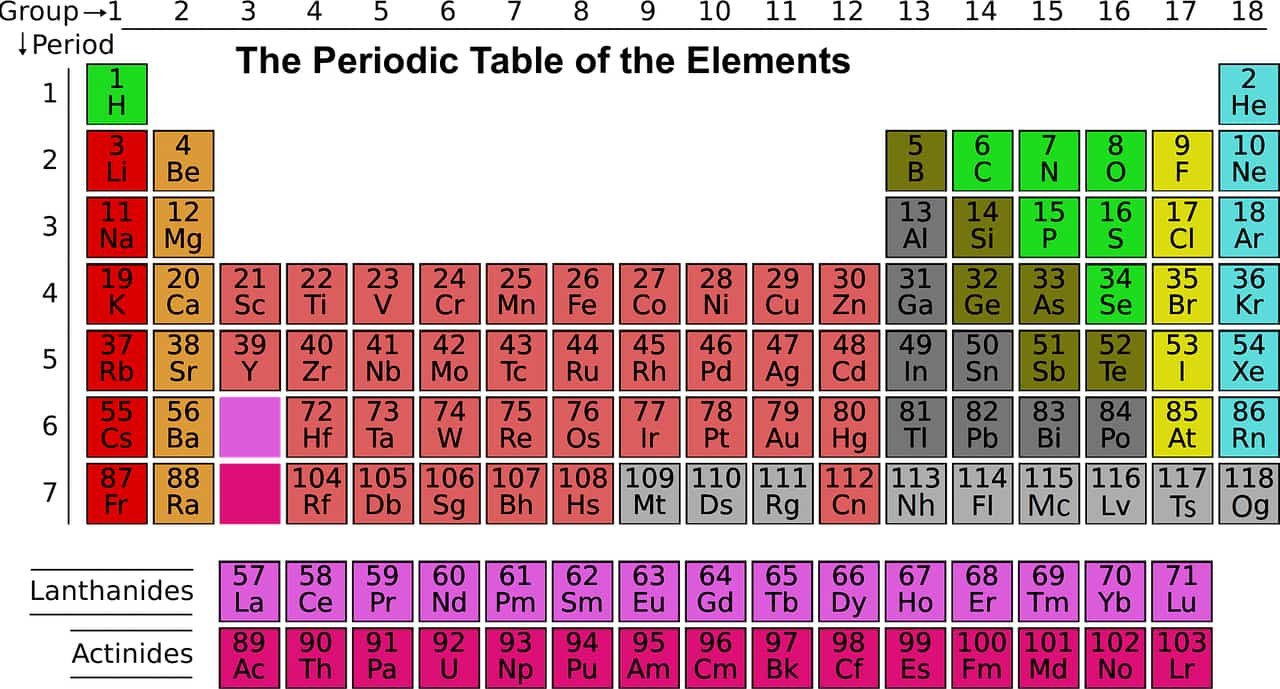
Sources: PinterPandai, Royal Society of Chemistry
Photo credit: Wikimedia Commons
Photo explanations: Pure barium in protective argon gas atmosphere.