Carbon is the chemical element with atomic number 6 and symbol C
Carbon has several types of allotropes, the most famous of which are graphite, diamond, and amorphous carbon. The physical properties of carbon vary depending on the type of allotrope. Carbon is the chemical element with atomic number 6 and symbol C. For example, diamond is transparent, whereas graphite is black and dull.
Diamond is one of the hardest materials in the world, while graphite is soft enough to leave its mark on paper.
Diamond has a very low electrical conductivity, while graphite is an excellent conductor of electricity. Under normal conditions, diamond has the highest thermal conductivity of known materials. All allotropes of carbon are solid under normal conditions, but graphite is the most thermodynamically stable allotrope of all.
Use of carbon
In the form of a free element, the carbon present in diamonds is used in jewelry and industry for its hardness. Graphite is used as a lubricant, to make crucibles and is also found in pencil leads.
In compound form, the applications of carbon are very numerous. CO2, for example, is used in particular in drinks (to add gas bubbles), in fire extinguishers, or as a refrigerant, in the form of dry ice, in the semiconductor industry. Carbon monoxide (CO) is used as a reducing gas in many industrial processes. Many metal carbides are valued for their high heat resistance. Carbon tetrachloride (CCl4) and carbon disulfide (CS2) are important solvents. Chlorofluorocarbon (CCl2F2), also called Freon R12, is a gas used in cooling systems.
This element has many uses, including jewelry decorations with diamonds or the black dye used for automobile rims or printer ink. Another form of carbon, graphite, is used at high temperatures for crucibles, dry and light cell vault electrodes, pencil tips and as a lubricant. Plant carbon, an amorphous form of carbon, is used as an absorbent gas and a bleaching agent.
Its compounds have a lot of uses. Carbon dioxide is used in the carbonation of beverages, in fire extinguishers and, in semiconductors, as a cooler (dry ice). Carbon monoxide is used as a reducing agent in many metallurgical processes.
Carbon tetrachloride and carbon disulfide are important industrial solvents. Freon is used in cooling systems. Calcium carbide is used to prepare acetylene; it is used for welding and cutting metals, as well as for the preparation of other organic compounds. Other metal carbides have important uses as heat resistance and metal cutters.
Dangers of carbon and its compounds
Pure carbon has low toxicity to humans and can be safely handled and even ingested in the form of graphite or charcoal. It is resistant to dissolution or chemical attack, even in the acidic contents of the digestive tract, for example.
In contrast, carbon disulfide CS2, although similar in structure to carbon dioxide, is a highly toxic liquid used as a solvent (rubber vulcanization).
The other carbon oxides are carbon monoxide CO, and the less common carbon suboxide C3O2. Carbon monoxide is a colorless, odorless gas formed by incomplete combustion of organic compounds or pure carbon (coal). Carbon monoxide binds more strongly than oxygen to blood hemoglobin to form carboxyhemoglobin, a stable compound. The result of this reaction is the poisoning of hemoglobin molecules, which can be fatal (see entry in question).
The CN– cyanide ion behaves chemically similar to a halide ion. Salts containing cyanide ion are highly toxic. Cyanogen, a gas of composition (CN)2, is also close to halogens.
With metals, carbon forms C4- carbides or C22- acetylides. Whatever happens, with an electronegativity of 2.5, carbon prefers to form covalent bonds. Some carbides are covalent lattices, like silicon carbide, SiC, which looks like diamond, and is moreover used for the size of these.
The toxicity of new allotropic forms of carbon (fullerenes, nanotubes, graphene) is now widely studied. In their native state, these nanostructures remain difficult to filter in the air and could constitute a danger that needs to be assessed. It should be noted that in the context of their use, these compounds are generally found dispersed in a solvent, or fixed on a solid substrate.
Forms of carbon
Graphite
The sp 2 -covalently hexagonally bonded carbon atoms form high-strength planes. The levels are only loosely bound by Van der Waals forces. Macroscopically, the cleavage dominates along the planar planes. Because the planes are so thin, their extraordinary strength is not apparent with graphite.
Because of this structure, graphite behaves very anisotropically : along the crystal planes, graphite is thermally and electrically very conductive, but heat conduction or charge transfer from crystal plane to crystal plane works relatively poorly.
Diamond
The sp 3 -covalently tetragonally bonded carbon atoms have no free electrons. The material is an insulator with a band gap of 5.45 eV that does not absorb visible light. The addition of foreign atoms creates states in the band gap and thus changes the electrical and optical properties. The yellowish tone of many natural diamonds is due to nitrogen, while boron-doped diamonds look bluish and are semiconducting. In the absence of air, the diamond transforms into graphite at temperatures around 1500 °C. It burns to carbon dioxide at around 700–800 °C.
Diamond is generally regarded as the metastable form of carbon under normal conditions (1 bar, 25 ° C) . However, based on recent research, this is no longer certain because
- the thermodynamic stability is only extrapolated to low PT conditions,
- in equilibrium studies the influence of the environment – small traces of impurities that are below the current detection limit can already have drastic effects on the equilibrium position of a reaction – was / is not taken into account and finally
- Experiments by Chinese scientists show that in a reaction between metallic sodium and magnesium carbonate, carbon and diamond coexist stably next to each other.
Read also: Diamond Grade | Carat, Clarity, Color and Cut
Lonsdaleit
Lonsdaleite, also known as the hexagonal diamond , is a very rare modification of the diamond. It occurs when graphite is converted into diamond by shock events, i.e. high pressure and high temperature such as impact events . The hexagonal character of the crystal structure is retained, however, in contrast to graphite, each carbon atom is covalently bonded to four others .
Chaoit
Chaoite is a very rare modification that crystallizes in the hexagonal crystal system similar to graphite, but with different lattice parameters and a slightly different crystal structure. Similar to Lonsdaleit, it is created by shock metamorphosis in graphitic gneiss.
Fullerenes
A regular hexagonal honeycomb pattern, as it is formed by the carbon atoms in the basal planes of graphite, is planar. If some hexagons are replaced by pentagons, curved surfaces are created which “roll up” to form closed bodies with certain relative arrangements of the five and six rings. Such structures are implemented in the fullerenes.
The sp 2 bonds are no longer in one plane, but form a spatially closed structure. The smallest possible structure consists only of pentagons and requires 20 carbon atoms, the corresponding body is a pentagon dodecahedron.
This simplest fullerene has so far only been mass spectrometrichas been proven. One of the most stable fullerenes consists of 60 carbon atoms and, apart from hexagons, only contains pentagons that have no edge in common with any other pentagon. The resulting pattern (truncated icosahedron, an Archimedean body) resembles the pattern on an (old-fashioned) soccer ball.
It is named Buckminster fullerene in honor of Richard Buckminster Fuller. The molecular “spheres” of the fullerenes are bound to one another via relatively weak van der Waals interactions, similar to the basal planes in graphite. In the meantime, a number of fullerenes of different sizes have been isolated and in some cases also crystallized; they can therefore be considered rea lModification (s) apply. Fullerenes are believed to be found in all soot, for example in the soot over candle flames.
Cyclo [18] carbon
1,3,5,7,9,11,13,15,17-Cyclooctadecanonain or Cyclo carbon is an allotropic modification of carbon with the empirical formula C 18 . The molecule is a homocyclic compound, the carbon atoms are alternately linked by single and triple bonds . It therefore belongs to the polyynes and the cyclocarbons and was the first cyclocarbon ever to be isolated in 2019 at temperatures just above absolute zero .
Other forms of carbon
Amorphous-carbon
In amorphous carbon (aC) the atoms are networked without long-range order. The material can be produced with almost any sp 2 : sp 3 hybridization ratios, with the material properties transitioning smoothly from those of graphite to those of diamond. In this case, the term diamond-like coating or diamond-like carbon (DLC) is often used in industry.
If the proportion of sp 3 hybridization is over 70%, one speaks of tetrahedral amorphous carbon (ta-C). This material is characterized by high electrical resistance, extreme hardness and optical transparency. The production can be done by means of PVD or PECVD-Methods are done. The material is deposited as a layer (amorphous carbon layer).
Carbon fibers
Carbon fibers consist of graphite-like sp 2 -bonded carbon. Isotropic fibers behave similarly to polycrystalline graphite and have only low strengths. Fiber mats and bundles are used for thermal seals. By stretching during manufacture, it is possible to align the basal planes along the fiber axis. High-strength fibers are obtained with properties that approximate the theoretical values of graphite along the basal planes. Anisotropic carbon fibers are light, extremely stiff and strong and are used in composite materials .
Glassy carbon
It is combined with those of a high-technological material of pure carbon, the vitreous and ceramic properties of the graphite. In contrast to graphite, vitreous carbon has a fullerene-like microstructure. This results in a wide variety of positive material properties. The conductivity is lower than that of graphite, for example.
Graphs
A graphite basal plane of sp 2– hybridized carbon is called a graph . The thin layers are obtained by chemically splitting graphite. Embedded in plastics, it is suitable as a starting material for new composite materials or for investigations into two-dimensional crystals. Research is also being carried out into applications in electronics .
Activated carbon
Careful graphitization of organic materials, such as coconut shells, leads to a porous carbon. The cavities are connected to one another like a sponge and form a very large inner surface. Activated carbon filters dissolved substances in low concentrations from liquids and can adsorb gases.
Soot
Carbon black is also made of graphite-based carbon. The purer the carbon black, the more clearly the properties of graphite emerge. Lamp or candle soot is heavily contaminated with organic compounds that prevent the formation of larger graphite associations.
Carbon-nanotubes
Another form of carbon are sp 2 -hybridized carbon atoms arranged in a cylindrical manner . Its geometry is created from a planar layer of graphite that is rolled up into a cylinder. The resulting tube can also be twisted, which changes the electrical properties. Several single-walled tubes can lie concentrically one inside the other, so that one speaks of multi-walled carbon nanotubes (MWCNT), in contrast to single-walled carbon nanotubes (SWCNT).
Carbon-nanofoam
Carbon nanofoam, an airgel , is a randomly oriented, network-like arrangement of carbon-graphite layers. It is similar to vitreous carbon, only with significantly larger interconnected cavities. Their average diameter is six to nine nanometers .
This is to be distinguished from carbon airgel (aerogel) , which consists of nanoparticles that have grown together. Its density is between 200 and 1000 kg / m3.
Aero graphite
Aerographite consists of a network of porous carbon tubes and is one of the lightest solids in the world with a density of 0.2 milligrams per cubic centimeter. Aerographite can be compressed by up to 95% and pulled apart again into its original shape.
Non-graphitic carbon
It consists of layers of hexagonally arranged, sp2 -hybridized carbon atoms. These layers are stacked almost parallel without any three-dimensional long-range order”. This material consists of stacks of graphene layers that are twisted and shifted against each other. This arrangement is also referred to as turbostratic.
The distance between the layers can differ significantly from the layer distance found in graphite. The microstructure analysis of the material is possible using WAXS, among other things, but standard methods such as the Scherrer equation can be used due to the wide and overlapping maxima caused by the significant disorder not be applied.
Carbine (pearl necklaces)
In 2016, it was possible to synthesize straight chains known as carbine , which consist of more than 6000 atoms, within double-walled nanotubes.
Q-carbon
Q-Carbon is a man-made diamond-like allotropic form of carbon that is described as ferromagnetic and harder than diamond.
Physical properties
Pure crystalline carbon exists in nature as diamond and graphite. Elemental carbon is inert and insoluble in water or organic solvents. At high temperatures, it oxidizes to form carbon monoxide and carbon dioxide.
We know of over a million compounds that have carbon bonded to hydrogen, as well as a large number of inorganic compounds that contain carbon. This element is bonded to hydrogen is found in particular in all plants and animals. Deposits of organic matter from living organisms fossilize to give various hydrocarbons. Carbonates are included in many minerals, gypsum and marble to name a few.
| Physical properties | |
|---|---|
| Phase at STP | solid |
| Sublimation point | 3915 K (3642 °C, 6588 °F) |
| Density (near r.t.) | amorphous: 1.8–2.1 g/cm3 graphite: 2.267 g/cm3 diamond: 3.515 g/cm3 |
| Triple point | 4600 K, 10,800 kPa |
| Heat of fusion | graphite: 117 kJ/mol |
| Molar heat capacity | graphite: 8.517 J/(mol·K) diamond: 6.155 J/(mol·K) |
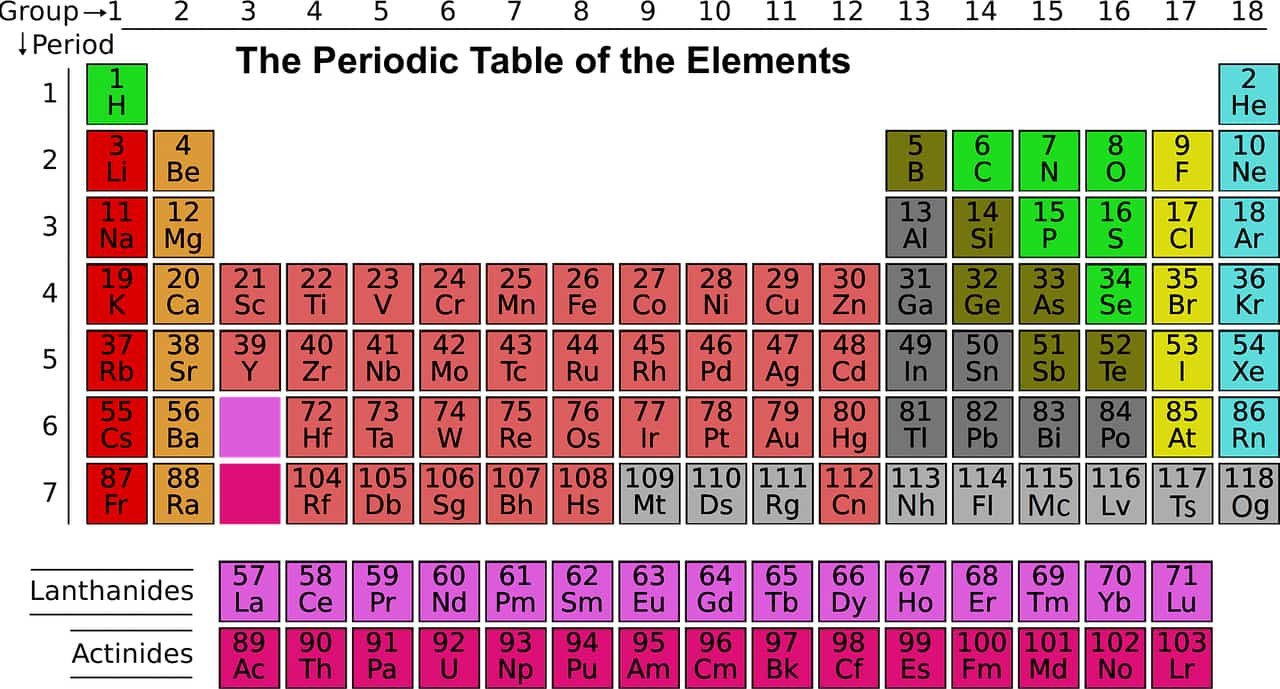
Carbon in the periodic table
| Atomic number (Z) | 6 |
|---|---|
| Group | group 14 (carbon group) |
| Period | period 2 |
| Block | p-block |
| Electron configuration | [He] 2s2 2p2 |
| Electrons per shell |
Atomic properties
| Atomic properties | |
|---|---|
| Oxidation states | −4, −3, −2, −1, 0, +1,+2, +3,+4 (a mildly acidic oxide) |
| Electronegativity | Pauling scale: 2.55 |
| Ionization energies |
|
| Covalent radius | sp3: 77 pm sp2: 73 pm sp: 69 pm |
| Van der Waals radius | 170 pm |
Other properties
| Other properties | |
|---|---|
| Natural occurrence | primordial |
| Crystal structure | graphite: simple hexagonal (black) |
| Crystal structure | diamond: face-centered diamond-cubic (clear) |
| Speed of sound thin rod | diamond: 18,350 m/s (at 20 °C) |
| Thermal expansion | diamond: 0.8 µm/(m·K) (at 25 °C) |
| Thermal conductivity | graphite: 119–165 W/(m·K) diamond: 900–2300 W/(m·K) |
| Electrical resistivity | graphite: 7.837 µΩ·m |
| Magnetic ordering | diamagnetic |
| Magnetic susceptibility | −5.9·10−6 (graph.) cm3/mol |
| Young’s modulus | diamond: 1050 GPa |
| Shear modulus | diamond: 478 GPa |
| Bulk modulus | diamond: 442 GPa |
| Poisson ratio | diamond: 0.1 |
| Mohs hardness | graphite: 1–2 diamond: 10 |
| CAS Number |
|
History
“Carbon” comes from the Latin carbo, which means “coal”. In the form of soot and charcoal, carbon has been known since prehistoric times. In 1772, Lavoisier demonstrated that diamond is a form of carbon. It will be the same for graphite in 1779 by Carl Wilhelm Scheele. It was again Lavoisier who listed carbon as an element in his works from 1789.
| History | |
|---|---|
| Discovery | Egyptians and Sumerians (3750 BCE) |
| Recognized as an element by | Antoine Lavoisier (1789) |
Main isotopes
Isotope Carbon-14, or radiocarbon, is a radioactive isotope of carbon with an atomic nucleus containing 6 protons and 8 neutrons. Its presence in organic materials is the basis of the radiocarbon dating method pioneered by Willard Libby and colleagues to date archaeological, geological and hydrogeological samples.
Isotope Carbon-12 is the more abundant of the two stable isotopes of carbon, amounting to 98.93% of the element carbon; its abundance is due to the triple-alpha process by which it is created in stars.
| Main isotopes | ||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||
Effects on health
It has a very low toxicity. The health risk data presented here are based on black carbon, non-elemental carbon exposures. Chronic inhalation exposure to black coal may cause temporary or permanent damage to the lungs and heart.
Pneumoconiosis has been found in workers working in coal production. Inflammation of hair follicles, and oral mucosal lesions have also been reported.
Carcinogenicity: Black anthrax has been listed by the International Agency for Research on Cancer (IARC: International Agency for Research on Cancer) in Group 3 (the agent is not classifiable as to its carcinogenicity to humans.)
Carbon-14 is one of the radionuclides involved in the atmospheric testing of nuclear weapons, which began in 1945 with a US test and ended in 1980 with a Chinese test. It is among the long-lived radionuclides which have produced and will continue to produce an increased risk of cancers for decades and centuries to come. It can also cross the placenta, become organically bound to cells, grow and therefore endanger the fetus.
Effects on the environment
No negative environmental effects have been reported.
Periodic Table of Elements | Complete List of Chemical Elements by Group, Name, Symbol, Color and Type
Sources: PinterPandai, Chemicool, Live Science, Royal Society of Chemistry
Photo source: Wikimedia Commons
Photo explanations:
Diamond and graphite shown side by side, for illustrating carbon. Scale is based on a rough approximation.
Diamond
Locality: South Africa
Size: 1.31 carats: 7 x 6 x 6 mm
An octahedral facetable crystal.
Graphite
Locality: El Cochi, Sonora, Mexico
Size: thumbnail, 2.5 x 2.4 x 1.2 cm
Crudely crystallized graphite, with a resinous luster.