Natural Occurrence of the Elements
In the periodic table there are 3 natural occurrence of the elements : Primordial, From decay and Synthetic.
The first 94 elements occur naturally; the remaining 24, americium to oganesson (95–118), occur only when synthesized in laboratories. Of the 94 naturally occurring elements, 83 are primordial and 11 occur only in decay chains of primordial elements. No element heavier than einsteinium (element 99) has ever been observed in macroscopic quantities in its pure form, nor has astatine (element 85); francium (element 87) has been only photographed in the form of light emitted from microscopic quantities (300,000 atoms).
Natural occurrence of the elements PRIMORDIAL
Primordial nuclides, also known as primordial isotopes, are nuclides found on Earth that have existed in their current form since before Earth was formed. Primordial nuclides were present in the interstellar medium from which the solar system was formed, and were formed in, or after, the Big Bang, by nucleosynthesis in stars and supernovae followed by mass ejection, by cosmic ray spallation, and potentially from other processes. They are the stable nuclides plus the long-lived fraction of radionuclides surviving in the primordial solar nebula through planet accretion until the present. Two hundred and eighty-six such nuclides are known.
List of 34 radioactive primordial nuclides and measured half-lives
These 34 primordial nuclides represent radioisotopes of 28 distinct chemical elements (cadmium, neodymium, samarium, tellurium, uranium, and xenon each have two primordial radioisotopes). The radionuclides are listed in order of stability, with the longest half-life beginning the list. These radionuclides in many cases are so nearly stable that they compete for abundance with stable isotopes of their respective elements. For three chemical elements, indium, tellurium, and rhenium, a very long-lived radioactive primordial nuclide is found in greater abundance than a stable nuclide.
The longest-lived radionuclide has a half-life of 2.2×1024 years, which is 160 trillion times the age of the Universe. Only four of these 34 nuclides have half-lives shorter than, or equal to, the age of the universe. Most of the remaining 30 have half-lives much longer. The shortest-lived primordial isotope, 235U, has a half-life of 703.8 million years, about one sixth of the age of the Earth and the Solar System.
| No. | Nuclide | Energy | Half- life (years) | Decay mode | Decay energy (MeV) | Approx. ratio half-life to age of universe |
|---|---|---|---|---|---|---|
| 253 | 128Te | 8.743261 | 2.2×1024 | 2 β− | 2.530 | 160 trillion |
| 254 | 124Xe | 8.778264 | 1.8×1022 | KK | 2.864 | 1 trillion |
| 255 | 78Kr | 9.022349 | 9.2×1021 | KK | 2.846 | 670 billion |
| 256 | 136Xe | 8.706805 | 2.165×1021 | 2 β− | 2.462 | 150 billion |
| 257 | 76Ge | 9.034656 | 1.8×1021 | 2 β− | 2.039 | 130 billion |
| 258 | 130Ba | 8.742574 | 1.2×1021 | KK | 2.620 | 90 billion |
| 259 | 82Se | 9.017596 | 1.1×1020 | 2 β− | 2.995 | 8 billion |
| 260 | 116Cd | 8.836146 | 3.102×1019 | 2 β− | 2.809 | 2 billion |
| 261 | 48Ca | 8.992452 | 2.301×1019 | 2 β− | 4.274, .0058 | 2 billion |
| 262 | 209Bi | 8.158689 | 2.01×1019 | α | 3.137 | 1 billion |
| 263 | 96Zr | 8.961359 | 2.0×1019 | 2 β− | 3.4 | 1 billion |
| 264 | 130Te | 8.766578 | 8.806×1018 | 2 β− | .868 | 600 million |
| 265 | 150Nd | 8.562594 | 7.905×1018 | 2 β− | 3.367 | 600 million |
| 266 | 100Mo | 8.933167 | 7.804×1018 | 2 β− | 3.035 | 600 million |
| 267 | 151Eu | 8.565759 | 5.004×1018 | α | 1.9644 | 300 million |
| 268 | 180W | 8.347127 | 1.801×1018 | α | 2.509 | 100 million |
| 269 | 50V | 9.055759 | 1.4×1017 | β+ or β− | 2.205, 1.038 | 10 million |
| 270 | 113Cd | 8.859372 | 7.7×1015 | β− | .321 | 600,000 |
| 271 | 148Sm | 8.607423 | 7.005×1015 | α | 1.986 | 500,000 |
| 272 | 144Nd | 8.652947 | 2.292×1015 | α | 1.905 | 200,000 |
| 273 | 186Os | 8.302508 | 2.002×1015 | α | 2.823 | 100,000 |
| 274 | 174Hf | 8.392287 | 2.002×1015 | α | 2.497 | 100,000 |
| 275 | 115In | 8.849910 | 4.4×1014 | β− | .499 | 30,000 |
| 276 | 152Gd | 8.562868 | 1.1×1014 | α | 2.203 | 8000 |
| 277 | 190Pt | 8.267764 | 6.5×1011 | α | 3.252 | 47 |
| 278 | 147Sm | 8.610593 | 1.061×1011 | α | 2.310 | 7.7 |
| 279 | 138La | 8.698320 | 1.021×1011 | K or β− | 1.737, 1.044 | 7.4 |
| 280 | 87Rb | 9.043718 | 4.972×1010 | β− | .283 | 3.6 |
| 281 | 187Re | 8.291732 | 4.122×1010 | β− | .0026 | 3 |
| 282 | 176Lu | 8.374665 | 3.764×1010 | β− | 1.193 | 2.7 |
| 283 | 232Th | 7.918533 | 1.405×1010 | α or SF | 4.083 | 1 |
| 284 | 238U | 7.872551 | 4.468×109 | α or SF or 2 β− | 4.270 | 0.3 |
| 285 | 40K | 8.909707 | 1.251×109 | β− or K or β+ | 1.311, 1.505, 1.505 | 0.09 |
| 286 | 235U | 7.897198 | 7.038×108 | α or SF | 4.679 | 0.05 |
Natural occurrence of the elements from DECAY (trace radioisotope)
A trace radioisotope is a radioisotope that occurs naturally in trace amounts (i.e. extremely small). Generally speaking, trace radioisotopes have half-lives that are short in comparison with the age of the Earth, since primordial nuclides tend to occur in larger than trace amounts. Trace radioisotopes are therefore present only because they are continually produced on Earth by natural processes. Natural processes which produce trace radioisotopes include cosmic ray bombardment of stable nuclides, ordinary alpha and beta decay of the long-lived heavy nuclides, thorium-232, uranium-238, and uranium-235, spontaneous fission of uranium-238, and nuclear transmutation reactions induced by natural radioactivity, such as the production of plutonium-239[1] and uranium-236 from neutron capture by natural uranium.
Elements
The elements that occur on Earth only in traces are listed below.
| Element name | Chemical Symbol |
|---|---|
| Technetium | Tc |
| Promethium | Pm |
| Polonium | Po |
| Astatine | At |
| Radon | Rn |
| Francium | Fr |
| Radium | Ra |
| Actinium | Ac |
| Protactinium | Pa |
| Neptunium | Np |
| Plutonium | Pu |
Isotopes of other elements (listnot exhaustive):
- Tritium
- Beryllium-7
- Beryllium-10
- Carbon-14
- Fluorine-18
- Sodium-22
- Sodium-24
- Magnesium-28
- Silicon-31
- Silicon-32
- Phosphorus-32
- Sulfur-35
- Sulfur-38
- Chlorine-34m
- Chlorine-36
- Chlorine-38
- Chlorine-39
- Argon-39
- Argon-42
- Calcium-41
- Iron-60
Natural occurrence of the elements SYNTHETIC
A synthetic element is one of 24 chemical elements that do not occur naturally on Earth: they have been created by human manipulation of fundamental particles in a nuclear reactor, a particle accelerator, or the explosion of an atomic bomb; thus, they are called “synthetic”, “artificial”, or “man-made”. The synthetic elements are those with atomic numbers 95–118, as shown in purple on the accompanying periodic table: these 24 elements were first created between 1944 and 2010. The mechanism for the creation of a synthetic element is to force additional protons onto the nucleus of an element with an atomic number lower than 95. All synthetic elements are unstable, but they decay at a widely varying rate: their half-lives range from 15.6 million years to a few hundred microseconds.
Five other elements that were created artificially—and thus initially considered to be synthetic—were later discovered to exist in nature in trace quantities. The first, technetium, was created in 1937. Plutonium, atomic number 94, first synthesized in 1940, is another such element. It is the element with the largest number of protons (and equivalent atomic number) to occur in nature, but it does so in such tiny quantities that it is far more practical to synthesize it. Plutonium is extremely well known due to its use in atomic bombs and nuclear reactors. No elements with an atomic number greater than 99 have any uses outside of scientific research, since they have extremely short half-lives, and thus have never been produced in large quantities.
List of synthetic elements
The following elements do not occur naturally on Earth. All are transuranium elements and have atomic numbers of 95 and higher.
| Element name | Chemical Symbol | Atomic Number | First definite synthesis |
|---|---|---|---|
| Americium | Am | 95 | 1944 |
| Curium | Cm | 96 | 1944 |
| Berkelium | Bk | 97 | 1949 |
| Californium | Cf | 98 | 1950 |
| Einsteinium | Es | 99 | 1952 |
| Fermium | Fm | 100 | 1952 |
| Mendelevium | Md | 101 | 1955 |
| Nobelium | No | 102 | 1966 |
| Lawrencium | Lr | 103 | 1961 |
| Rutherfordium | Rf | 104 | 1966 (USSR), 1969 (US) * |
| Dubnium | Db | 105 | 1968 (USSR), 1970 (US) * |
| Seaborgium | Sg | 106 | 1974 |
| Bohrium | Bh | 107 | 1981 |
| Hassium | Hs | 108 | 1984 |
| Meitnerium | Mt | 109 | 1982 |
| Darmstadtium | Ds | 110 | 1994 |
| Roentgenium | Rg | 111 | 1994 |
| Copernicium | Cn | 112 | 1996 |
| Nihonium | Nh | 113 | 2003–4 |
| Flerovium | Fl | 114 | 1999 |
| Moscovium | Mc | 115 | 2003 |
| Livermorium | Lv | 116 | 2000 |
| Tennessine | Ts | 117 | 2010 |
| Oganesson | Og | 118 | 2002 |
| * Shared credit for discovery. | |||
Other elements usually produced through synthesis
All elements with atomic numbers 1 through 94 occur naturally at least in trace quantities, but the following elements are often produced through synthesis. Technetium, promethium, astatine, neptunium, and plutonium were discovered through synthesis before being found in nature.
| Element name | Chemical Symbol | Atomic Number | First definite discovery |
|---|---|---|---|
| Technetium | Tc | 43 | 1937 |
| Promethium | Pm | 61 | 1945 |
| Polonium | Po | 84 | 1898 |
| Astatine | At | 85 | 1940 |
| Francium | Fr | 87 | 1939 |
| Actinium | Ac | 89 | 1902 |
| Protactinium | Pa | 91 | 1913 |
| Neptunium | Np | 93 | 1940 |
| Plutonium | Pu | 94 | 1940 |
Periodic Table of Elements | Complete List of Chemical Elements by Group, Name, Symbol, Color and Type
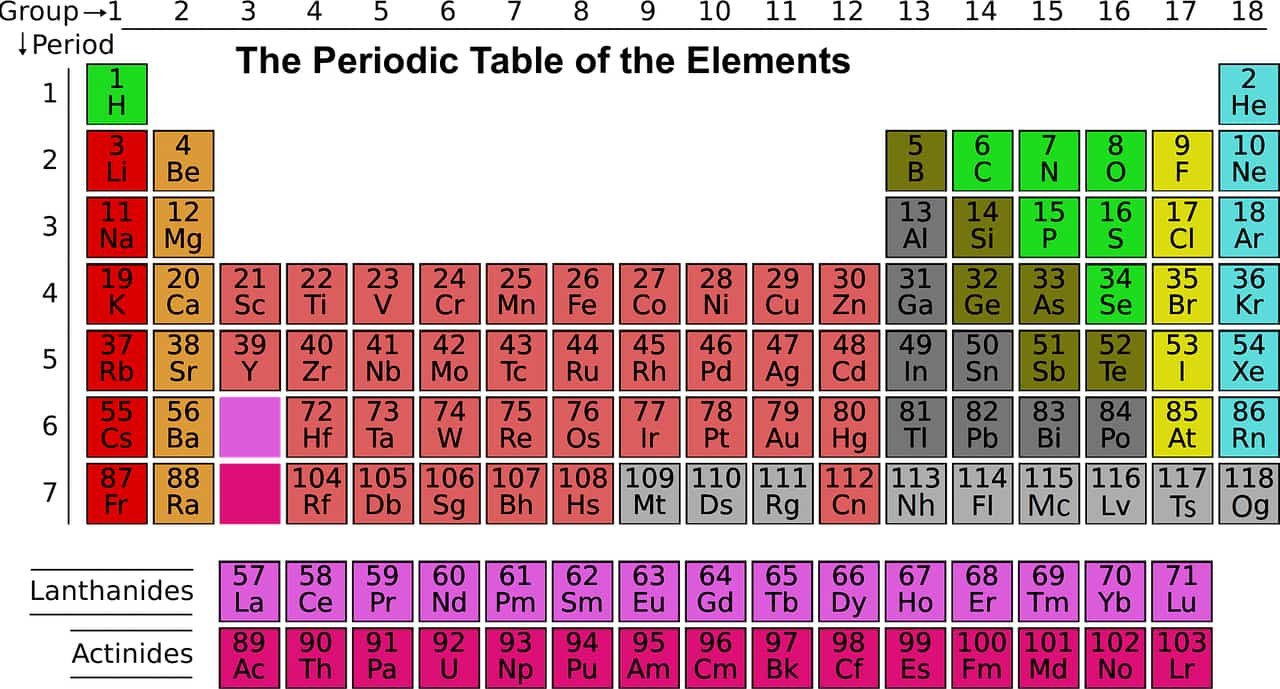
Sources: Royal Society of Chemistry, Web MD, Live Science
Photo credit: Wikimedia Commons


