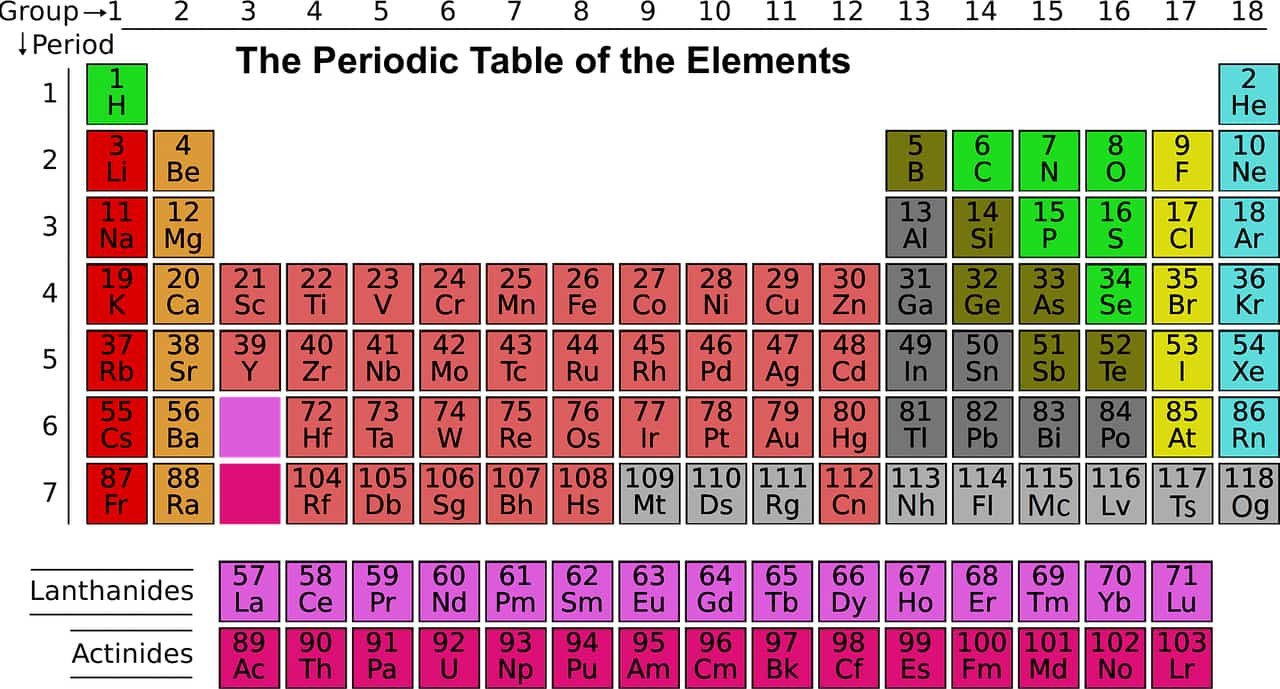
Beryllium is a chemical element with the symbol Be and atomic number 4
In the periodic table, Beryllium the first representative of the alkaline earth metals. The name beryllium comes from the Greek βήρυλλος (beryllos) which designated aquamarine or emerald.
A bivalent element, beryllium is an alkaline earth metal with a steel gray appearance. It is light, fragile and toxic.
It was discovered as a component of the mineral beryl by Louis-Nicolas Vauquelin as early as 1798 and was initially called glucine because of the sweet taste of the isolated beryllium-containing compounds (e.g. hydroxide). Elemental beryllium was first produced in 1828 by Friedrich Wöhler and independently of that by Antoine Bussy.
Uses
- Used as an alloying agent in the manufacture of beryllium copper. (Be able to absorb a lot of heat). Beryllium-copper alloys are used in a variety of uses due to their electrical and thermal conductivity, high strength and hardness, nonmagnetic properties, as well as corrosion and fatigue resistance. These uses include the manufacture of: molds, spot welding electrodes, springs, sparkless electronic equipment and electrical connectors.
- Due to its toughness, lightness, and dimensional stability over a wide temperature range, copper-beryllium alloys are used in the aerospace and defense industry as a light structuring material in high-speed aircraft, guided shells, aircraft and communications satellites.
- Thin strips of beryllium are used in conjunction with X-ray scans to exclude visible light and allow only X-rays to be detected.
- Its uses as insulation and external contact material in electronics, as well as its incorporation into silicone greases have also been abandoned, due to very significant health risks.
- In the field of X-ray lithography, beryllium is used for the manufacture of microscopic integrated circuits.
Due to the low heat absorption of neutrons, the nuclear power industry uses this metal in nuclear reactors as a neutron reflector and moderator. - Used in the manufacture of gyroscopes, various computer tools, watch springs and equipment that require lightness, rigidity and dimensional stability.
- Beryllium oxide is very useful in a variety of applications requiring a good heat conductor, and high strength and hardness, as well as a high melting point, thereby acting as an electrical barrier.
Beryllium alloys were used at one time in fluorescent lamps, but that use has been discontinued as exposed workers are in danger of beriliosis. - Nuclear field: mixed with an alpha emitter, such as americium, or high energy gamma if used as a source of long-lived neutrons necessary for reactor operation.
- In geomorphology and paleoseismology, the 10Be isotope, created by cosmic rays, is used for cosmogenic isotope dating of surfaces or for the determination of erosion rates.
- Other uses of its crystalline properties: X-ray window, for example window of an X-ray tube or of an X-ray detector: the window isolates the interior of the device from the environment.
Toxicity and Health Issues of Beryllium
Presentation – Ecotoxicology
Beryllium is a very toxic, non-radioactive metal. It is classified among the most toxic elements like arsenic (As), cadmium (Cd), chromium (Cr), lead (Pb), thallium (Tl) and mercury (Hg). Beryllium acts as a carcinogenic poison, affecting cell membranes and binding to certain regulatory proteins in cells. Beryllium may remain detectable in urine for up to 10 years after exposure. It is classified as a category 1 carcinogen by the European Union and is therefore in France subject to the CMR 2001-97 decree of February 1, 2001 (which applies to any exposure to beryllium).
Beryllium is ecotoxic (and in particular carcinogenic). It is the smallest of the metal cations. And if it seems relatively little mobile in water with neutral or alkaline pH, it is on the other hand in soils or environments that are naturally acidic (frequent in a large part of the world) or made acidic by humans.
The low natural abundance of beryllium (3 × 10-4%) means that it does not pose any particular environmental problem, but it can be concentrated in coals and in granite rocks such as pegmatites in the form of several minerals: bertrandite, beryl … It then circulates with bioavailability and a still poorly evaluated propensity to concentrate in certain organs or in certain species23,24. The thermal uses of coal injected a significant quantity of it into the atmosphere, the fallout of which enriched the environments around industrial and urban sites using coal (often associated with acid rain, especially for coals with a high sulfur content) .
There are controversies over its use in dentistry in dentures.
Detection of beryllium in the human body at very high doses is always associated with harmful effects (of varying severity). Beryllium is:
- Alkaline phosphatase inhibitor;
- Hepatic adenosine triphosphate inhibitor;
- DNA synthesis inhibitor.
This results in health effects, which are classified into several categories:
Non-carcinogenic effects
The inhalation of “large” concentrations of beryllium (more than 1 mg per cubic meter of air), or prolonged inhalation (more than ten years) even of low doses, can cause a disease called chronic disease of the body. beryllium or beryllium (or CBD for Chronic Beryllium Disease). This disease affects the lungs, has many points in common with pneumonia, and can progress to severe cardiorespiratory failure.
Animal model: While ingestion of beryllium by the human body has not shown direct and harmful effects on the stomach and intestine, ingestion of beryllium by animals results in the presence of lesions in the these organs.
Sensitization: Some people become hypersensitive to beryllium (they develop an allergic reaction to this element). In a few people, direct exposure to beryllium (skin contact) has caused skin damage with or without granulomatosis and respiratory tract inflammation.
Carcinogenic effects
Several studies have been done on the increase in the number of deaths from lung cancer among people employed in factories using beryllium.
Read also: Why Do Many People Get Cancer?
Sources of contamination of the human body
The contamination of the human body by beryllium occurs mainly by 4 ways:
1. By inhalation of air containing beryllium particles (dust, fumes, vapors, nanoparticles from abrasion).
2. By ingestion of contaminated food and drinking water.
3. Following the corrosion of the beryllium alloys present in the mouth.
4. By skin and transcutaneous contamination, very sensitizing “The contribution of skin absorption is increasingly suspected in the development of sensitization”.
Air contamination
Beryllium can be very harmful when inhaled. In fact, there is a great correlation between the level of beryllium in human urine and that in the air, which proves that the contamination in the human body is not only due to water pollution but also to the atmosphere.
Contamination by drinking water
It can be found in natural waters and industrial effluents in trace amounts. In general, the concentration of beryllium in natural water and wastewater varies between 0.1 and 500 μg / L, but when this concentration exceeds 0.2 μg / L, we begin to speak of an environmental problem.
Contamination by dentures
In contact with saliva, any alloy containing beryllium corrodes and releases ions which diffuse into the surrounding tissues and are partly ingested. Corrosion is all the stronger with these alloys as beryllium, a very reactive metal, reacts in the presence of any other metal. Chronic intoxication which results from the permanent diffusion of beryllium ions in the body is a factor of dysregulation of the immune system, in particular in people allergic (or sensitized to beryllium following prolonged contact).
Dentists (milling) and dental prosthetists are also exposed to it through their work (in 1990, 50% of dental prosthetists used a beryllium alloy).
Notable features
Beryllium has the highest melting point of all light metals. It is fragile, but lighter and six times stronger than aluminum. Its ductility is approximately a third greater than that of steel.
It has excellent thermal conductivity, is non-magnetic and is resistant to concentrated nitric acid. It is highly permeable to X-rays, and releases neutrons when struck by alpha particles, such as those emitted by radium or polonium.
Under normal temperature and pressure conditions, beryllium is resistant to oxidation when exposed to air. A thin layer of oxide forms which gives it its ability to scratch glass.
In nature, it is mainly found in the form of complex oxides or aluminosilicates called beryls, the best-known valuable representatives of which are emerald and aquamarine.
It is exploited from around thirty ores (especially bertrandite and beryl). The world’s main mines are in the United States, China and Mozambique. None are open in Europe.
Beryllium in the periodic table
| Atomic number (Z) | 4 |
|---|---|
| Group | group 2 (alkaline earth metals) |
| Period | period 2 |
| Block | s-block |
| Electron configuration | [He] 2s2 |
| Electrons per shell | 2, 2 |
Physical properties
| Physical properties | |
|---|---|
| Phase at STP | solid |
| Melting point | 1560 K (1287 °C, 2349 °F) |
| Boiling point | 2742 K (2469 °C, 4476 °F) |
| Density (near r.t.) | 1.85 g/cm3 |
| when liquid (at m.p.) | 1.690 g/cm3 |
| Critical point | 5205 K, MPa (extrapolated) |
| Heat of fusion | 12.2 kJ/mol |
| Heat of vaporization | 292 kJ/mol |
| Molar heat capacity | 16.443 J/(mol·K) |
Vapor pressure
| P (Pa) | 1 | 10 | 100 | 1 k | 10 k | 100 k |
|---|---|---|---|---|---|---|
| at T (K) | 1462 | 1608 | 1791 | 2023 | 2327 | 2742 |
Atomic properties
| Atomic properties | |
|---|---|
| Oxidation states | 0, +1, +2 (an amphoteric oxide) |
| Electronegativity | Pauling scale: 1.57 |
| Ionization energies |
|
| Atomic radius | empirical: 112 pm |
| Covalent radius | 96±3 pm |
| Van der Waals radius | 153 pm |
Other properties
| Other properties | |
|---|---|
| Natural occurrence | primordial |
| Crystal structure | hexagonal close-packed (hcp) |
| Speed of sound thin rod | 12,890 m/s (at r.t.)[4] |
| Thermal expansion | 11.3 µm/(m·K) (at 25 °C) |
| Thermal conductivity | 200 W/(m·K) |
| Electrical resistivity | 36 nΩ·m (at 20 °C) |
| Magnetic ordering | diamagnetic |
| Magnetic susceptibility | −9.0·10−6 cm3/mol |
| Young’s modulus | 287 GPa |
| Shear modulus | 132 GPa |
| Bulk modulus | 130 GPa |
| Poisson ratio | 0.032 |
| Mohs hardness | 5.5 |
| Vickers hardness | 1670 MPa |
| Brinell hardness | 590–1320 MPa |
| CAS Number | 7440-41-7 |
History
| History | |
|---|---|
| Discovery | Louis Nicolas Vauquelin (1798) |
| First isolation | Friedrich Wöhler & Antoine Bussy (1828) |
Main isotopes of beryllium
Beryllium has 12 known isotopes, with a mass number varying between 5 and 16. Only 9Be is stable and represents almost all of natural beryllium. Two of the radioisotopes of beryllium have been detected in nature: 10Be with a half-life of 1.39 million years, and 7Be with a half-life of 53.22 days; both are cosmogenic nuclides created by the interaction of cosmic rays with the nuclei of atoms in the air. The other radioisotopes have very short half-lives and are only detectable in the instruments that were used to create them artificially.
| Main isotopes of beryllium | |||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||
Periodic Table of Elements | Complete List of Chemical Elements by Group, Name, Symbol, Color and Type
Information: Cleverly Smart is not a substitute for a doctor. Always consult a doctor to treat your health condition.
Sources: PinterPandai, Researchgate, CDC, Merck KGaA
Photo source: Wikimedia Commons
Photo explanations: Beryllium, >99 % pure, crystalline big fragment >140 g.