Gamma Radiation (Gamma γ Rays)
Often denoted by the Greek letter gamma, ). Gamma rays are an energized form of electromagnetic radiation produced by radioactivity/other nuclear/subatomic processes such as electron-positron annihilation.
What are gamma rays?
Gamma radiation or rays are a type of wave on the electromagnetic spectrum. Of all the waves in the spectrum, gamma rays have the shortest wavelength and the highest energy.
Did you know?
Physicist Ernest Rutherford coined the term “gamma ray”. His work on gamma rays, carried out at McGill University, won him the Nobel Prize in chemistry.
Gamma rays are produced in many situations.
Most often, they are found in space as gamma-ray bursts. These are the most powerful explosions in the Universe since the Big Bang.
On Earth, lightning and nuclear explosions produce gamma rays. It is also possible to create them using a laser. For example, the Gemini laser at the Appleton Rutherford Laboratory, located in the United Kingdom, produces gamma rays.
Easy Definition of Gamma Rays
Gamma rays may sound foreign to us because of their form in the form of rays (radiation) and also because gamma rays can only be found in nuclear processes. Maybe what we often hear is gamma rays for the treatment of cancer, tumors and other diseases.
Benefits of Gamma Rays
Kills bacteria.
Gamma rays are radiation that has the same high energy as X-rays. Which with high energy can damage the cells of living things, therefore it is not surprising that gamma rays can kill bacteria. That’s why gamma rays can be used to:
a. Sterilizing food and drinks
b. Sterilize doctor’s equipment before performing surgery.
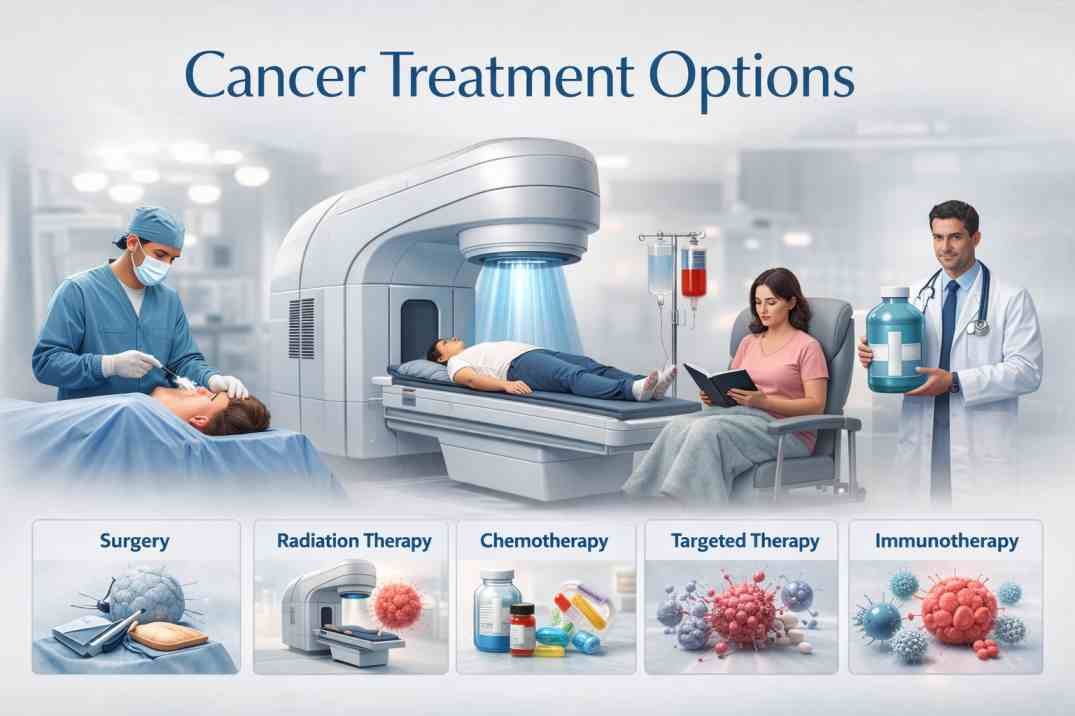
Cure tumors, cancer, and other disorders.
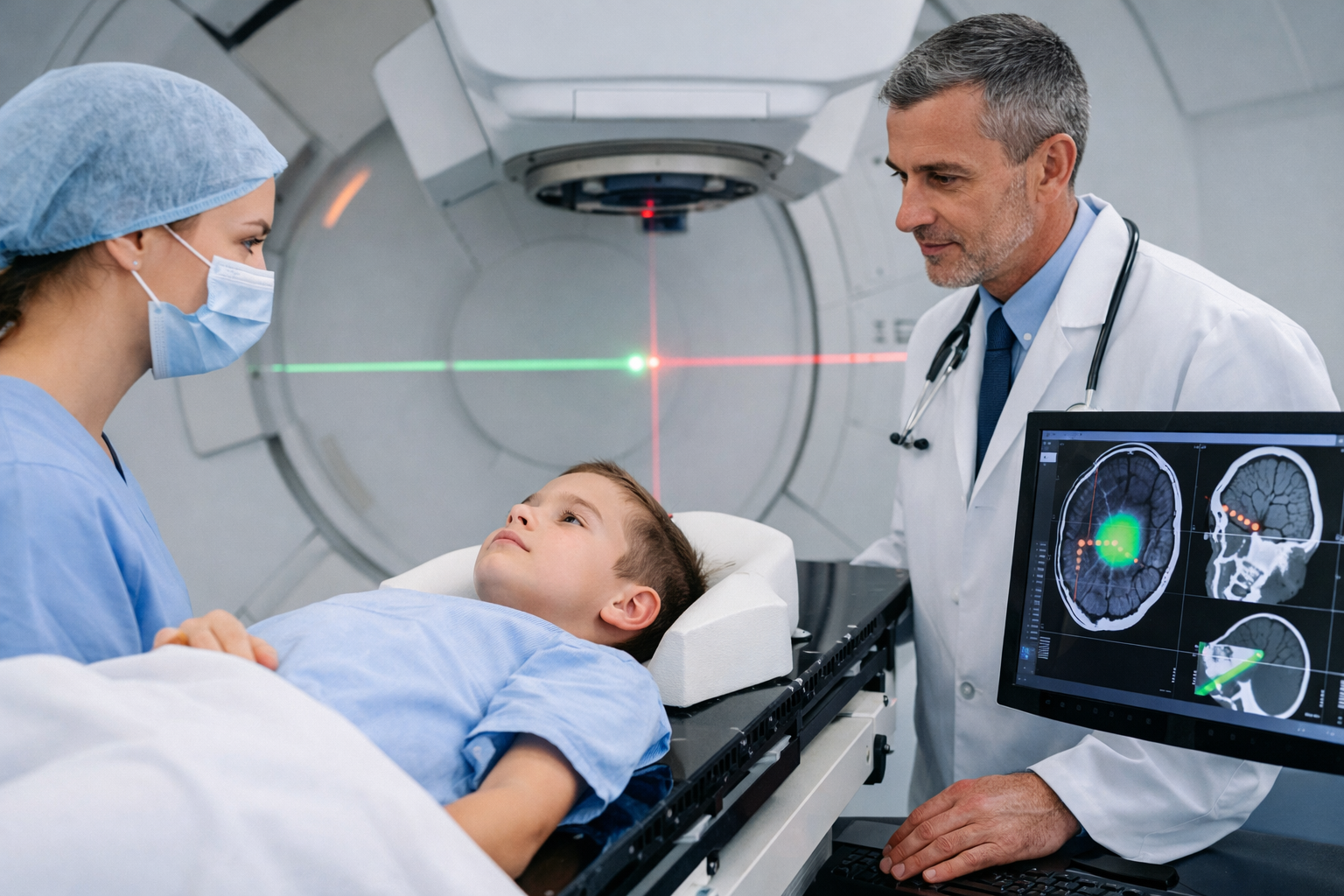
Gamma rays can actually be used to kill cancer cells and tumors and other disorders because gamma rays can destroy these cells. This therapy is called the gamma knife.
Knowing the study / research for: knowing the structure of metals, knowing superior seeds, to make radio isotopes and many more.
The Danger of Gamma Radiation
Damaging satellites and the atmosphere.
Gamma rays from outer space can actually damage satellites and the atmosphere. This happens because of the very strong energy of gamma rays. Even so, we can breathe a sigh of relief because according to scientists these gamma bursts are rare. That is about 10 thousand – 1 million years. The last occurred in the 8th century.
Causes the greatest death in the event of a nuclear war.
In the explosion of a nuclear weapon a lot of radioactive material is created. However, Gamma rays from a nuclear fallout are likely to cause the largest number of deaths from the use of nuclear weapons in a nuclear war. An effective fallout protection will reduce human exposure 1000 times.
Are gamma rays dangerous?
Large doses of radiation can be dangerous to humans. They can even be fatal.
In science fiction films, gamma radiation often creates mutant creatures, leading viewers to believe that the radiation is dangerous and should be avoided.
In reality, we are surrounded by sources of radiation. This occurs naturally in space and in the earth’s crust. Radiation is therefore present in the food we eat and in the air we breathe. Radiation is also produced by humans. For example, it is emitted by lasers.
But even taken together, not all of these radiation sources are dangerous. It would take about 750 times the radiation to hurt you.
Gamma Ray Properties
The properties possessed by gamma rays are high energy so that their penetrating power is very strong. Gamma rays have the strongest penetrating power compared to the waves that fall into the electromagnetic wave group. Gamma rays can penetrate iron plates up to several centimeters thick.
Gamma Ray Source
Gamma rays are formed due to other nuclear or subatomic processes such as electron-positron annihilation. Gamma rays cannot be found anywhere because they can only occur as a result of other nuclear and subatomic processes. These rays can form when:
- Star explosion or supernova.
- Nuclear bomb explosion.
- Radioactive material.
- A star sucked in by a black hole.
- Gamma ray therapy.
- The energy bubble at the center of the Milky Way galaxy.
- Know the metal structure.
- Making radio isotopes.
Gamma radiation
Gamma radiation is a highly penetrating electromagnetic radiation, which arises from the spontaneous transformation (“decay”) of the atomic nuclei of many naturally occurring or artificially occurring radioactive nuclides.
The name comes from the division of ionizing radiation from radioactive decay into alpha radiation, beta radiation and gamma radiation with an increased ability to penetrate matter. Alpha and beta radiation are composed of charged particles and therefore interact much more strongly with matter than photons or quanta of uncharged gamma radiation. Correspondingly, the latter has a much higher penetration capacity.
Different types of electromagnetic spectrum (gamma radiation)
In a broader sense, gamma radiation is used to denote electromagnetic radiation with a quantum energy above about 200 keV, regardless of how it originates.
This corresponds to a wavelength shorter than 0.005 nm (5 pm). In this general sense, the term is used specifically when the process by which the radiation is generated is unknown (e.g. in astronomy) or irrelevant to a particular task (e.g. in radiation protection), but it must be stated that the higher energy compared to X-rays (approx. 100 eV to 300 keV).
Use of gamma rays
Gamma rays provide information about some of the most energetic phenomena in the universe; However, they are mostly absorbed by the Earth’s atmosphere. Instruments aboard high-altitude balloons and satellite missions, such as the Fermi Gamma-ray Space Telescope, provide our only view of the universe in gamma rays.
Gamma-induced molecular changes can also be used to change the properties of semi-precious stones, and are often used to convert white topaz to blue topaz.
Non-contact industrial sensors commonly use gamma radiation sources in the refining, mining, chemical, food, soap and detergent, and pulp and paper industries, for level, density, and thickness. Gamma ray sensors are also used to measure liquid levels in the water and oil industry. Typically, these use the isotope Co-60 or Cs-137 as the radiation source.
In the US, gamma-ray detectors are being used as part of the Container Security Initiative (CSI). This machine is advertised to be able to scan 30 containers per hour.
Gamma radiation is often used to kill living organisms, in a process called irradiation. These applications include sterilization of medical equipment (as an alternative to autoclaving or chemical means), removal of spoilage-causing bacteria from many foods and prevention of germination of fruits and vegetables to preserve freshness and taste.
Apart from their cancer-causing properties, gamma rays are also used to treat certain types of cancer, as these rays also kill cancer cells. In a procedure called gamma knife surgery, several concentrated gamma rays are directed at the growth to kill cancer cells. The beams are directed from different angles to concentrate radiation on the growth while minimizing damage to the surrounding tissue.
Gamma rays are also used for diagnostic purposes in nuclear medicine in imaging techniques. A number of different gamma emitting radioisotopes are used. For example, in a PET scan, a radio-labeled sugar called fluorodeoxyglucose emits a positron that is annihilated by electrons, producing a gamma-ray pair that highlights cancer because cancers often have a higher metabolic rate than surrounding tissue. The most common gamma emitter used in medical applications is the nuclear isomer technetium-99m which emits gamma rays in the same energy range as diagnostic X-rays. When this radionuclide tracer is administered to a patient, a gamma camera can be used to form an image of the radioisotope distribution by detecting the emitted gamma radiation. Depending on which molecules have been labeled with the tracer, the technique can be used to diagnose various conditions (for example, spread of cancer to bone via bone scan).
Discovery history
The first historically discovered source of gamma rays was a radioactive decay process called gamma decay. In this type of decay, the excited nucleus emits gamma rays immediately after formation (this is now understood to be a nuclear isomeric transition, although inhibited gamma decay with a measured and longer half-life can also occur). Paul Villard, a French chemist and physicist, discovered gamma radiation in 1900 while studying radiation emitted by radio.
Villard knew that the radiation was much stronger than the previously described types of radio beam radiation, such as beta rays., first observed as “radioactivity” by Henri Becquerel in 1896, and alpha rays, discovered to be a less penetrating form of radiation by Rutherford, in 1896. 1899. However, Villard did not consider when naming them that they were a different type of elementary radiation. Villard was recognized in 1903 by Ernest Rutherford as a fundamentally different type of rays, and were also referred to as “gamma rays”, by analogy with alpha and beta rays. which he had differentiated into 1899.
The rays emitted by radioactive elements are named according to their power to penetrate various materials, using the first three letters of the Greek alphabet: alpha rays, which penetrate the least, followed by beta rays and gamma rays, which are the most penetrating. Rutherford also realized that gamma rays were not deflected (or at least not easily deflected) by magnetic fields, another property that distinguished them from alpha and beta rays.
Gamma rays were originally thought to be particles with mass, such as alpha and beta rays. Rutherford believed that they could be very fast beta particles, but the inability to deflect them by a magnetic field showed that they had no charge.
In 1914, gamma rays were observed bouncing off the glass surface, proving that it was electromagnetic radiation.
Rutherford and his colleague Edward Andrade measured the wavelengths of radio gamma rays, and found that they were similar to X-rays, but with a shorter wavelength and (hence) a higher frequency. This was eventually recognized by giving them more energy per photon as well, soon after the latter term became generally accepted. Gamma decay was then understood as the emission of a single gamma photon.
Sources: PinterPandai, National Center for Biotechnology Information (NCBI), Health Physics Society, Ministry of the Environment Government of Japan, Occupational Safety & Health Administration, National Center for Biotechnology Information (NCBI)